Our quality infrastructure aligns with cGMP and ISO 22716 cosmetic requirements while using technology to enforce compliance at every step of production.
Talk to Our TeamQuality is enforced by process, documentation, and system design - not memory or manual checks.
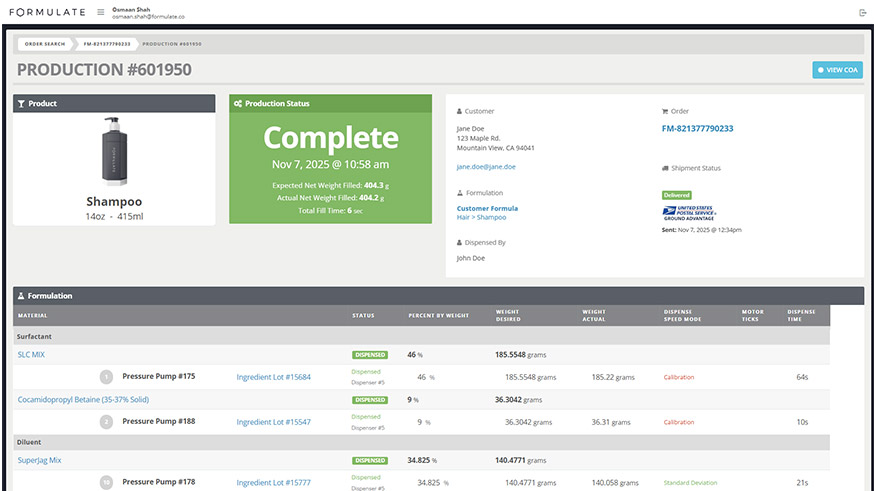
Our operations follow globally recognized cosmetic manufacturing frameworks that guide how we manage facilities, equipment, personnel, documentation, and production. These standards inform how we structure workflows, manage records, and maintain consistent manufacturing outcomes.
Current good manufacturing practices
Global standard for cosmetics good manufacturing practices.
Launching a product requires more than a working formula - it requires alignment with regulatory expectations across documentation, ingredients, labeling, and manufacturing practices.
Our quality control testing is integrated directly into production workflows, ensuring consistency, traceability, and early detection of issues before product release.
By aligning with cGMP and ISO 22716 standards - and digitizing quality and regulatory workflows - our platform provides a manufacturing foundation built for compliance, consistency, and long-term growth.